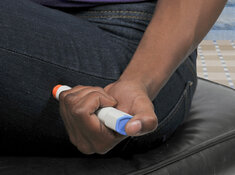
Continuous insulin delivery system company Insulet Corp. (PODD:NASDAQ), which incorporates tubeless insulin pump technology into its Omnipod® product line, today announced "it has received clearance from the U.S. Food and Drug Administration (FDA) for its Omnipod® 5 Automated Insulin Delivery System (Omnipod 5) for individuals aged six years and older with type 1 diabetes."
The company reported that its "Omnipod 5 is the first tubeless automated insulin delivery (AID) system that integrates with the Dexcom G6 Continuous Glucose Monitoring (CGM) System and a compatible smartphone to automatically adjust insulin and help protect against highs and lows."
The firm advised that Omnipod 5 helps simplify daily life for those being monitored and treated for diabetes as it allows individuals to manage their glucose levels without the use of multiple daily injections, tubes, or fingersticks.
Insulet Corp.'s President and CEO Shacey Petrovic remarked, "Omnipod 5 is a life-changing technology that we believe will revolutionize the market and the lives of people with diabetes…We are incredibly proud of this simple-to-use, elegant system, designed to deliver unmatched freedom and to greatly simplify insulin management and improve glucose control for our users."
The Omnipod 5 System is comprised of a tubeless Pod that features SmartAdjust™ technology that interfaces with Dexcom's G6 CGM. The system is operated remotely through either the Omnipod 5 mobile app or the firm's Omnipod 5 Controller. The device tracks glucose levels continuously in five-minute intervals and is then able to predict with high certainty what glucose levels will be one hour into the future. The system then will act to pause, increase or decreases the amount of insulin based upon the user's medically prescribed glucose target, thus alleviating highs and lows.
Kevin Sayer, Chairman, President and CEO of Dexcom Inc. (DXCM:NASDAQ) stated, "As the pioneer of integrated CGM, we are excited to see our years of collaborative work with Insulet culminate into the first and only FDA-cleared tubeless automated insulin delivery system…Omnipod 5 combines the accuracy and unmatched user experience of the Dexcom G6 CGM with the simplicity of tubeless insulin delivery to offer people with diabetes a revolutionary new way to optimize time in range."
The company stated it plans to launch and distribute Omnipod 5 via its existing pharmacy channels. The firm said it will first offer the Omnipod 5 in a limited market release to insure a best-in-class product experience when a wider rollout is implemented.
Insulet mentioned that "Omnipod 5 was recently selected as a CES® 2022 Innovation Award honoree in two categories: Health and Wellness and Wearable Technologies."
Insulet is based in Acton, Mass. and is focused on improving lives of those diagnosed with diabetes and other conditions. The firm's Omnipod Insulin Management System and platform offers a unique alternative to traditional insulin delivery methods. The company's wearable disposable pods can administer up to three days of non-stop, intermittent insulin without the need for patients to regularly inject themselves. The company believes that the Omnipod technology may also prove useful in other applications to deliver other non-insulin related subcutaneous drugs.
Insulet Corp. started the day with a market cap of around $13.4 billion with approximately 68.97 million shares outstanding and a short interest of about 3.9%. PODD shares opened 13% higher today at $220.00 (+$25.20, +12.94%) over yesterday's $194.80 closing price. The stock has traded today between $211.20 and $228.58 per share and is currently trading at $225.00 (+$30.20, +15.50%).
Disclosure:
1) Stephen Hytha compiled this article for Streetwise Reports LLC and provides services to Streetwise Reports as an independent contractor. He or members of his household own securities of the following companies mentioned in the article: None. He or members of his household are paid by the following companies mentioned in this article: None.
2) The following companies mentioned in this article are billboard sponsors of Streetwise Reports: None. Click here for important disclosures about sponsor fees.
3) Comments and opinions expressed are those of the specific experts and not of Streetwise Reports or its officers. The information provided above is for informational purposes only and is not a recommendation to buy or sell any security.
4) The article does not constitute investment advice. Each reader is encouraged to consult with his or her individual financial professional and any action a reader takes as a result of information presented here is his or her own responsibility. By opening this page, each reader accepts and agrees to Streetwise Reports' terms of use and full legal disclaimer. This article is not a solicitation for investment. Streetwise Reports does not render general or specific investment advice and the information on Streetwise Reports should not be considered a recommendation to buy or sell any security. Streetwise Reports does not endorse or recommend the business, products, services or securities of any company mentioned on Streetwise Reports.
5) From time to time, Streetwise Reports LLC and its directors, officers, employees or members of their families, as well as persons interviewed for articles and interviews on the site, may have a long or short position in securities mentioned. Directors, officers, employees or members of their immediate families are prohibited from making purchases and/or sales of those securities in the open market or otherwise from the time of the decision to publish an article until three business days after the publication of the article. The foregoing prohibition does not apply to articles that in substance only restate previously published company releases.
6) This article does not constitute medical advice. Officers, employees and contributors to Streetwise Reports are not licensed medical professionals. Readers should always contact their healthcare professionals for medical advice.