Nu-Med Plus Inc. (NUMD:OTCMKTS) is closely watching two clinical studies underway in which nitric oxide gas inhalation is being evaluated as a treatment for mild to moderate COVID-19, the company announced in a news release.
"We are watching the clinical trials closely for positive results in treatment of COVID-19," Nu-Med Plus CEO Jeff Robins said in the release. "Our patent-pending, nitric oxide disposable delivery device may possibly make a difference in helping to combat this disease and others in the future."
Nu-Med is a biotech firm that researches and develops delivery devices and medical applications for nitric oxide.
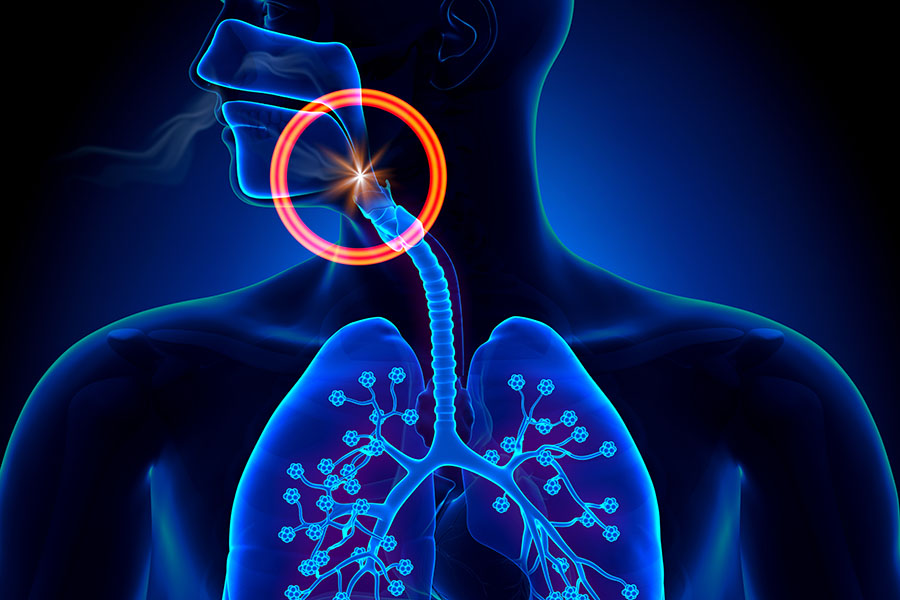
The studies, a collaboration of China's Xijing Hospital and Massachusetts General Hospital, commenced in early March. The hypothesis behind them is that because inhaled nitric oxide was used previously as a treatment component for patients infected with and demonstrating pulmonary complications from severe acute respiratory syndrome (SARS), it could play a role in COVID-19 therapy as well. This is because the two coronaviruses share genomic characteristics.
Nu-Med also is in communication with the U.S. Food and Drug Administration about guidance on how to accelerate getting several of its products onto the market. Nu-Med and the Veterans Administration and Nu-Med and Guangzhou, China, city officials are considering possible research on inhaled nitric oxide and COVID-19.
Jeff Robins, CEO of Nu-Med Plus, commented, "Scientists continue to research and recognize the impressive healing power of inhaled nitric oxide to help alleviate a diverse array of global health conditions. While nitric oxide has not been specifically studied or used to treat COVID-19, in vitro it has demonstrated an inhibitory effect on the replication cycle of SARS-CoV providing a strong basis for the clinical trials. We are watching the clinical trials closely for positive results in treatment of COVID-19. Our patent pending nitic oxide disposable delivery device may possibly make a difference in helping to combat this disease and others in the future."
[NLINSERT]Disclosure:
1) Doresa Banning compiled this article for Streetwise Reports LLC and provides services to Streetwise Reports as an independent contractor. She or members of her household own securities of the following companies mentioned in the article: None. She or members of her household are paid by the following companies mentioned in this article: None.
2) The following companies mentioned in this article are billboard sponsors of Streetwise Reports: None. Click here for important disclosures about sponsor fees.
3) Comments and opinions expressed are those of the specific experts and not of Streetwise Reports or its officers. The information provided above is for informational purposes only and is not a recommendation to buy or sell any security.
4) The article does not constitute investment advice. Each reader is encouraged to consult with his or her individual financial professional and any action a reader takes as a result of information presented here is his or her own responsibility. By opening this page, each reader accepts and agrees to Streetwise Reports' terms of use and full legal disclaimer. This article is not a solicitation for investment. Streetwise Reports does not render general or specific investment advice and the information on Streetwise Reports should not be considered a recommendation to buy or sell any security. Streetwise Reports does not endorse or recommend the business, products, services or securities of any company mentioned on Streetwise Reports.
5) From time to time, Streetwise Reports LLC and its directors, officers, employees or members of their families, as well as persons interviewed for articles and interviews on the site, may have a long or short position in securities mentioned. Directors, officers, employees or members of their immediate families are prohibited from making purchases and/or sales of those securities in the open market or otherwise from the time of the interview or the decision to write an article until three business days after the publication of the interview or article. The foregoing prohibition does not apply to articles that in substance only restate previously published company releases.