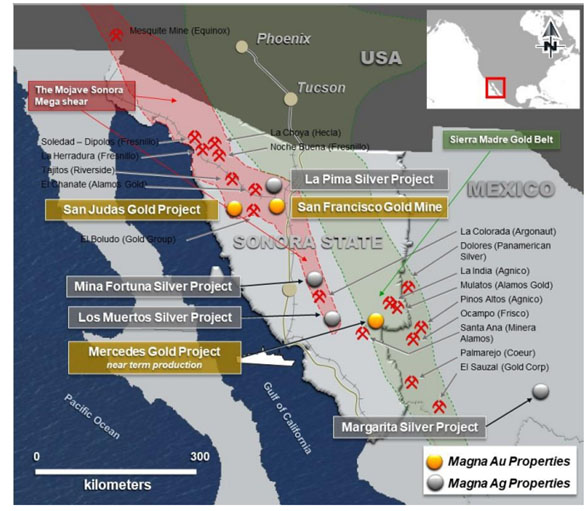
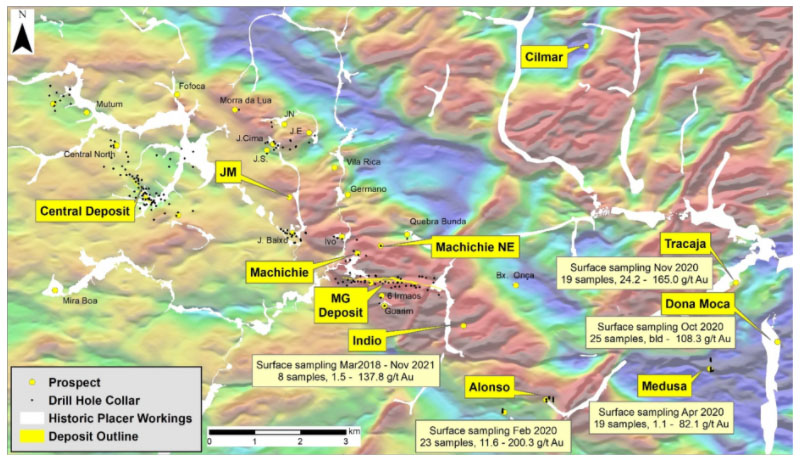
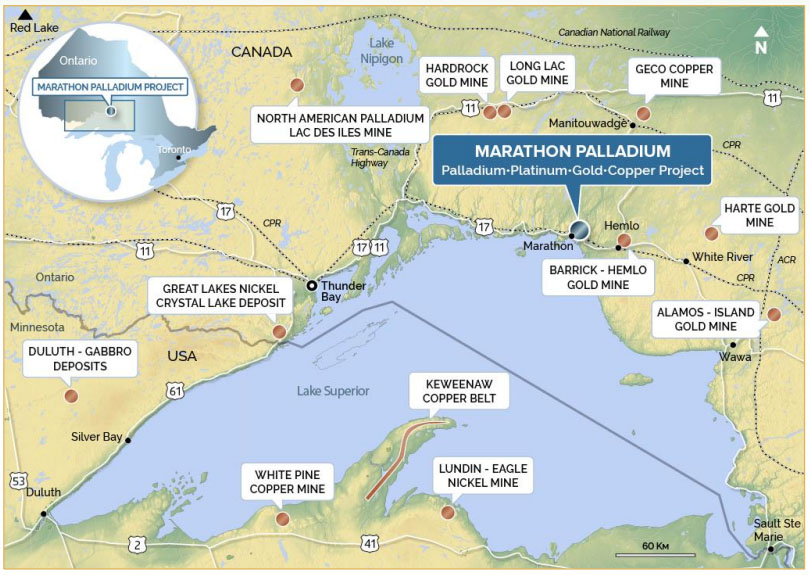
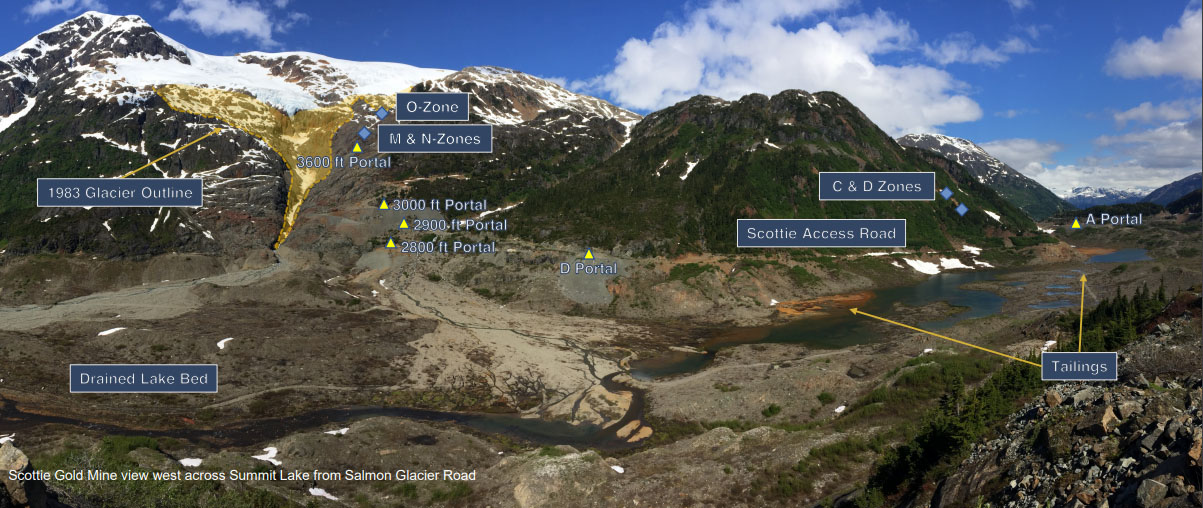
In a news release, Avivagen Inc. (VIV:TSX.V; VIVXF:OTCQB) announced that in light of COVID-19, it is fast tracking the commercial launch of its antibiotic-free OxC-beta product as an immune boosting supplement for humans.
"The seriousness of the COVID-19 pandemic tells us that we have a duty to bring our product forward to try and assist in the global fight to contain the spread of and impact of COVID-19," CEO Kym Anthony said in the release. "Longer term, we know that the benefits of our patent-protected OxC-beta product will help countless people face daily challenges by having a primed innate immune system ready to fight pathogens and helping keep undesirable inflammation at bay."
To accelerate OxC-beta's commercial debut as a supplement for humans in the U.S., Avivagen hired Bloom Burton & Co.'s consulting group to assist with the regulatory path, strategy and timing.
Already, Avivagen launched OxC-beta first as an addition to livestock feeds and also as a supplement for companion animals.
The life sciences firm has been studying the transmission, symptoms and progression of COVID-19 and comparing those data to its body of scientific work on what OxC-beta achieves and how.
Those data show that the product "supports and primes the innate immune system while also dampening chronic, overzealous inflammatory responses, elements that could prove beneficial to those with heightened risk of infection with COVID-19, seasonal flu, the common cold and future emerging diseases caused by novel pathogens," the release noted. For instance, a study in bovine respiratory disease, which can cause respiratory failure and death in calves, showed that treatment with OxC-beta of Holstein calves helped resolve lung inflammation.
According to the company, OxC-beta's mechanism of action involves boosting the body's number of pathogen-sensing receptors, including Toll-like receptor-2 and receptor-4, which surveil for harmful pathogens. It also increases the expression of genes involved in pathogen defense, including IFNGR1/CD119, TLR2, TLR4, CD14 and LY96/MD2.
Read what other experts are saying about:
Disclosure:
1) Doresa Banning compiled this article for Streetwise Reports LLC and provides services to Streetwise Reports as an independent contractor. She or members of her household own securities of the following companies mentioned in the article: None. She or members of her household are paid by the following companies mentioned in this article: None.
2) The following company mentioned in this article is a billboard sponsor of Streetwise Reports: Avivagen. Click here for important disclosures about sponsor fees.
3) Comments and opinions expressed are those of the specific experts and not of Streetwise Reports or its officers. The information provided above is for informational purposes only and is not a recommendation to buy or sell any security.
4) The article does not constitute investment advice. Each reader is encouraged to consult with his or her individual financial professional and any action a reader takes as a result of information presented here is his or her own responsibility. By opening this page, each reader accepts and agrees to Streetwise Reports' terms of use and full legal disclaimer. This article is not a solicitation for investment. Streetwise Reports does not render general or specific investment advice and the information on Streetwise Reports should not be considered a recommendation to buy or sell any security. Streetwise Reports does not endorse or recommend the business, products, services or securities of any company mentioned on Streetwise Reports.
5) From time to time, Streetwise Reports LLC and its directors, officers, employees or members of their families, as well as persons interviewed for articles and interviews on the site, may have a long or short position in securities mentioned. Directors, officers, employees or members of their immediate families are prohibited from making purchases and/or sales of those securities in the open market or otherwise from the time of the interview or the decision to write an article until three business days after the publication of the interview or article. The foregoing prohibition does not apply to articles that in substance only restate previously published company releases. As of the date of this article, officers and/or employees of Streetwise Reports LLC (including members of their household) own securities of Avivagen, a company mentioned in this article.