BrainStorm Cell Therapeutics Inc. (BCLI:NASDAQ) today announced publication of data from its Phase 1/2 and Phase 2a dose escalation trials in the Journal of the American Medical Association (JAMA) Neurology.
The news was announced in a press release and via conference call from San Francisco, where investors and executives from biotech and pharmaceutical companies across the globe have gathered for the 2016 JP Morgan Healthcare Conference and the 2016 Biotech Showcase.
BrainStorm CEO Chaim Lebovits and the paper's lead author, Dr. Dimitrios Karussis of Hadassah Medical Center in Jerusalem, discussed the JAMA publication, elaborated on the company's future plans, and took questions from investors during the call.
The JAMA article, said Lebovits, lends "credibility" to what his company is developing.
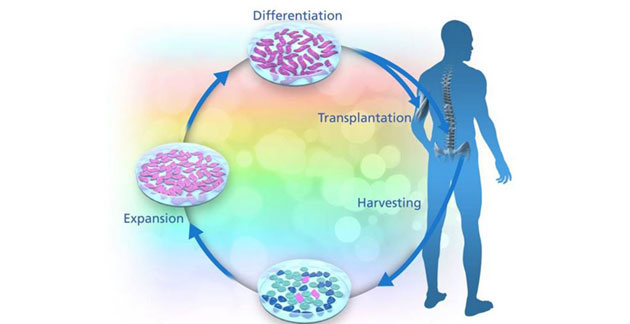
BrainStorm's NurOwn cells, an autologous stem cell therapy in which mesenchymal stem cells from bone marrow are differentiated into neuron-supporting cells (MSC-NTF), are being studied in amyotrophic lateral sclerosis (ALS) and other neurological diseases. The JAMA publication discusses the "outcome of the first-in-man Phase 1/2 study and Phase 2 dose escalation study with NurOwn in ALS."
NurOwn treatment demonstrated "excellent" safety, as well as what Karussis described as "revolutionary" data that is the first to show significant improvement in slowing the course of progression of the fatal and incurable disease.
According to Karussis, 90% of patients treated in the Phase 2a Israeli trial were "responders," and demonstrated a 25% improvement in course of progression, which is "considered clinically meaningful." The improvements were measured for a variety of functions, including ALS Functional Rating Scale-Revised (ALS-FRS-R) score and respiratory function. There was also "more objective" evidence of efficacy via electromyography and MRI. He anticipates that the ongoing Phase 2 trial in the U.S. will confirm the safety and efficacy data.
BrainStorm is also presently conducting a Phase 2 trial with NurOwn in the United States. This trial, which is randomized, double-blind, and placebo-controlled, will evaluate safety and efficacy in 48 patients with ALS.
The company is also in the planning stages for a new Phase 2 trial involving multiple doses of NurOwn cells, since data show the effects of the cells are strongest in the first three months after treatment, and then taper off. But some patients show benefit a year after being dosed, Lebovits said. The multidose trial will also be a collaboration with the Hadassah Medical Center.
Though the studies discussed in the JAMA paper were not placebo-controlled, Karussis asserted in the conference call that the placebo effect likely did not come into play because there were two study arms. In one arm, the cells were injected intramuscularly, and in the other they were injected intrathecally (into the space around the spinal cord). Patients who received intrathecal administration demonstrated the most marked improvement, Karussis said.
The doctor and CEO also fielded questions from parents of patients who praised the results they saw when their loved ones were treated with NurOwn cells. They called on the company to provide further dosing via compassionate use, if possible. But Lebovits and Karussis both demurred, citing regulatory restrictions. The regulatory process has its faults, Lebovits observed on the call, but there's "no better system out there."
He and Karussis both pledged to do everything in their power, within those regulatory confines, to further compassionate or accelerated use of the therapy.
Read what other experts are saying about:
DISCLOSURE:
1) Tracy Salcedo prepared this article for Streetwise Reports LLC, publisher of The Gold Report, The Energy Report and The Life Sciences Report, and provides services to Streetwise Reports as an employee. She owns, or her family owns, shares of the company mentioned in this interview: None.
2) BrainStorm Cell Therapeutics Inc. is a sponsor of Streetwise Reports.
3) The interview does not constitute investment advice. Each reader is encouraged to consult with his or her individual financial professional and any action a reader takes as a result of information presented here is his or her own responsibility. By opening this page, each reader accepts and agrees to Streetwise Reports' terms of use and full legal disclaimer.
4) From time to time, Streetwise Reports LLC and its directors, officers, employees or members of their families, as well as persons interviewed for articles and interviews on the site, may have a long or short position in securities mentioned. Directors, officers, employees or members of their families are prohibited from making purchases and/or sales of those securities in the open market or otherwise during the up-to-four-week interval from the time of the interview until after it publishes.