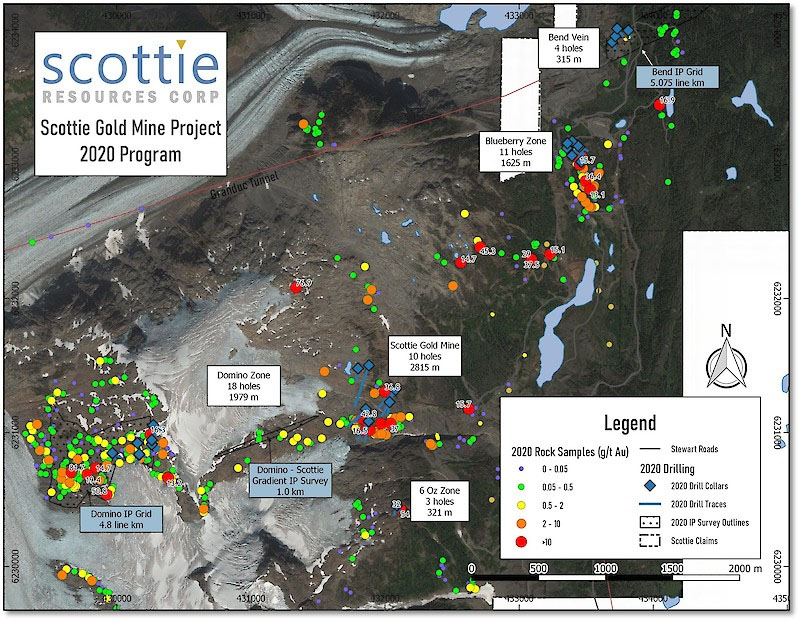
Lithium batteries have become the portable powerhouses of modern society. But good as they are, lithium batteries are not up to the demanding task of powering the next generation of electric vehicles. They just don't have enough juice or the ability to release it quickly over and over again.
However, the way forward is clear: find a way to improve the cathode's specific capacity while maintaining all the other characteristics that batteries require, such as a decent energy efficiency and a good cycle life.
Today, Hailiang Wang and buddies at Stanford University say they've achieved a significant step towards this goal using sulphur as the cathode material of choice.
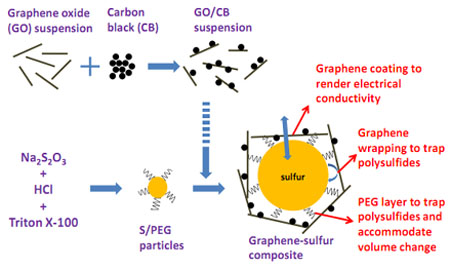
Chemists have known for many years that sulphur has potential: it has a theoretical specific capacity of 1672 mAh/g. Wang and co say they've largely overcome sulfur's conducting problems using a few clever nanoengineering techniques. Their trick is to create submicron sulphur particles and coat them in a kind of plastic called polyethyleneglycol or PEG. This traps polysulphides and prevents them from washing away.
Next, Wang and co wrap the coated sulphur particles in a graphene cage. The interaction between carbon and sulphur renders the particles electrically conducting and also supports the particles as they swell and shrink during each charging cycle.
The result is a cathode that retains a specific capacity of more than 600 mAh/g over 100 charging cycles.
That's impressive. Such a cathode would immediately lead to rechargeable lithium batteries with a much higher energy density than is possible today.
The next step then is to create a working battery out of this stuff. If it all works out, your next car could be powered by Li-S batteries.