In 1946 New York City, the Anti-Trust Division of the Department of Justice (DOJ) filed a complaint against Inco and its wholly owned U.S. subsidiary, International Nickel Co. Inc. Canada's Inco, at the time, owned 90% of the world's nickel ore and supplied 90% of U.S. nickel needs.
The charges brought against it include:
- conspiracy to prevent competition in the nickel industry;
- fixing prices; and
- making cartel agreements with I.G. Farbenindustrie, A.G. and two French companies to prevent competition and peg prices in the world market.
How ironic that, in 2010, the U.S. did not have any active nickel mines. Nickel has a very interesting history and is still extremely important in everyday functions of modern economies.
In 1751, Axel Fredrik Cronstedt of Sweden attempted to extract copper from the mineral Kupfernickel (today called niccolite). To his surprise, instead of copper he got a silvery white metal he started calling nickel. The name nickel comes from the German language and means Old Nick, which is a name Germans use for the devilso, nickel is "Old Nicks copper," or the "Devil's copper."
In 1913, Harry Brearly an English scientist, was the first to produce stainless steel when he accidentally discovered the addition of chromium makes stainless steel stainless. More than 10.5% chromium must be added to allow the outside protective oxide film to form on the steel, which provides corrosion resistance and gives stainless steel its bright, silvery appearance. The more chromium added, the greater the corrosion resistance.
Nickel is an important alloying addition in nearly two-thirds of the stainless steel produced today. Its primary function is to stabilize the austenitic (face-centered cubic crystal) structure of the steel. Normal carbon steel will, on cooling, transform from an austenite structure to a mixture of ferrite and cementite. When added to stainless steel, nickel stops this transformation, keeping the material fully austenite on cooling. Austenitic stainless steels have high ductility, low-yield stress and high-tensile strength when compared to carbon steel. Other examples of metals with austenitic structure include aluminum and copper.
The minimum amount of nickel that will stabilize the austenitic structure is 8%. That's the percentage present in the most-commonly used grade of stainless steelType 304, which is 18% chromium, 8% nickel and is known as "18/8." This composition was one of the first to be developed, it was used for chemical plants and also to clad the Chrysler Building in New York City.
Today's Use of Nickel
Almost 40% of annual nickel use is in nonferrous alloys (mixed with metals other than steel) and super alloys (metal mixtures designed to withstand extremely high temperatures and/or pressures or have high electrical conductivity). Nickel is present in over 3,000 different alloys used in more than 250,000 end-use applications.
Nickel is used as a coating on other metals to slow down corrosion, for the production of coins, as a catalyst for certain chemical reactions and as a colorantnickel is added to glass to give it a green color.
Rechargeable nickel-hydride batteries are used for cellular phones, video cameras, and other electronic devices. Nickel-cadmium batteries are used to power cordless tools and appliances.
It is estimated there is roughly 130 million tons of land-based resources averaging at least one percent nickel available in identified deposits. Nickel-bearing deposits come in two types:
- Nickel sulphide deposits are formed from the precipitation of nickel minerals by hydrothermal fluids. These sulfide deposits are also called magmatic sulfide deposits and are found in Australia, Canada, Russia and South Africa and are typically associated with copper and platinum group metals.
- Nickel laterite Deposits
are formed from weathering of ultramafic rocks and are usually operated as open pit mines. They are located principally in Western Australia, New Caledonia, Indonesia, Colombia, Cuba, Venezuela, Brazil and the Dominican Republic.
The trend of future nickel production is changing because of the current lack of high quality nickel sulfide exploration targetsnickel laterites are most likely to be developed as the world's future primary nickel sources.
Three countries dominate the top-three spots, in terms of nickel deposits:
- Russia is the world's leading country for nickel production and Russian mining giant Norilsk Nickel is the world's largest producer. Most of the countries nickel production (an amazing one-fifth of global production) is from Norilskthe largest nickel sulfide deposit in the world.
- Canada is the world's second largest nickel producing country. Most of the countries nickel currently comes from the Thompson Nickel Belt in Manitoba, the Sudbury Basin of Ontario, and the Ungava peninsula of Quebec.
- Vale SA, a Brazilian company and the world's second largest mining company by market value, said its total nickel output will be cut by about five percent this year as it repairs a damaged furnace in Canada. The company, whose goal is to become the world's top nickel producer in 2011, said on February 7th 2011 the No. 2 furnace at its Copper Cliff nickel plant in Sudbury, Ontario would be shut down for at least 16 weeks.
Australia is the world's third-most important producer of nickel. The country primarily exports its nickel products to Europe, Japan and the U.S.
Nickel Sulphide Deposits
Magmas (magma is a mixture of molten rock, volatiles and solids that is found beneath the surface of the EarthLava is the extrusive equivalent of magma) originate in the upper mantle and contain small amounts of nickel, copper and PGE. As the magmas ascend through the crust they cool as they encounter the colder crustal rocks.
If the original sulfur (S) content of the magma is sufficient, or if S is added from crustal wall rocks, a sulphide liquid forms as droplets dispersed throughout the magma. Because the partition coefficients of nickel, copper, iron and Platinum Group Elements (PGE) favor sulphide liquid these elements transfer into the sulphide droplets in the magma. The sulphide droplets sink toward the base of the magma because of their greater density and form sulphide concentrations. On further cooling, the sulphide liquid crystallizes to form the ore deposits that contain these metals.
Currently, the majority of today's nickel is produced from sulphide deposits, as it is easier and cheaper to mine and process than lateritic ore. However known sulphide deposits are getting depleted, ore grades are falling and new discoveries are scarce.
There are two main types of nickel sulphide deposits. In the first, Ni-Cu sulphide deposits, nickel (Ni) and copper (Cu) are the main economic commoditiescopper may be either a coproduct or byproduct, and cobalt (Co), Platinum Group Elements (PGE) and gold (Au) are the usual byproducts.
The second type of deposit is mined exclusively for PGE's with the other associated metals being byproducts.
Nickel sulphide deposits can occur as individual sulphide bodies but groups of deposits may occur in areas or belts ten's, even hundreds of kilometers long. Such groups of deposits are known as districts. Two giant Ni-Cu districts stand out above all the rest in the world: Sudbury Ontario, and Norilsk-Talnakh, Russia.
The most important platinum-rich PGE district in the world is the Bushveld Complex, South Africa. The Fraser Institute mining survey ranks the attractiveness of mining investment destinations. This year's survey shows South Africa has fallen from 27/47 places in the 2003 period to 67/79 this year. South Africa is now just above Zimbabwe and the Democratic Republic of the Congo in the Fraser Institute survey rankings. The second PGE district in importance is the Norilsk-Talnakh district, which is exceptionally Palladium (Pd) rich as a byproduct of its Ni-Cu ores.
Nickel Laterite Deposits
Nickel laterite deposits were first discovered in 1864 by French civil engineer Jules Garnier in New Caledoniacommercial production started in 1875. New Caledonia's laterites were the world's largest source of nickel until Sudbury Ontario's sulphide deposits started production in 1905 and totally dominated global production for the next 70 years.
Roughly 60%, or 84 Mt., of global available nickel is in laterite deposita deposit in which weathering of ultramafic rocks has taken place. The initial nickel content is strongly enriched in the course of lateritization and, under tropical conditions, fresh rock weathers very quickly. Some metals may be leached away by the weathering process but others, such as aluminum, iron and nickel can remain.
Typically, nickel laterite deposits are very large tonnage, low-grade deposits located close to the surface. They tend to be tabular and flat covering many square kilometers. They are most often in the range of 20 Mt. and upward, with some examples approaching 1 billion tons (Bt.) of material.
Laterite deposits usually contain both an upper dark red limonite (higher in iron and lower in nickel, magnesium and silica) and lower bright green saprolite zone (higher nickel, magnesium and silica but lower iron content). Due to the different quantities of iron, magnesium and silica in each zone they must be processed differently to cost-effectively retrieve the nickel.
HPAL Technology
Most nickel-sulfide deposits have traditionally been processed by concentration through a froth-flotation process, followed by pyrometallurgical extraction
Laterite saprolite (higher nickel, magnesium and silica but lower iron content) ore bodies are processed with standard pyrometallurgical technology. However, a laterite-limonite zone is higher in iron and lower in nickel, magnesium and silica, which means using High Pressure Acid Leaching (HPAL) technology. HPAL involves processing ore in a sulphuric acid leach at temperatures up to 270º C and pressures up to 600 PSI to extract the nickel and cobalt from the iron-rich ore; the pressure leaching is done in titanium-lined autoclaves.
Counter-current decantation is used to separate the solids and liquids. Separating and purifying the nickel/cobalt solution is done by solvent extraction and electrowinning.
Heap Leach
Heap leach treatment of nickel laterites is primarily used for ore types that are high oxide and low clay contentclay content needs to be low enough to allow percolation of the acid through the heap.
Ore is ground, agglomerated and maybe mixed with clay free rock for better permeability. The ore is then stacked on impermeable plastic membranes and acid is percolated over the heap for up to four months. Between 60% and 70% of the nickel/cobalt is liberated into the acid solutionthis acid solution is then neutralized with limestone and nickel/cobalt hydroxide is produced, which is then sent to a smelter for refining.
Conclusion
Unlike gold, there is no "emotional" component to the value of nickel. It isn't hoarded in safes, given as wedding presents or dangled around women's necks. Instead, you find it in buildings, autos and bridges.
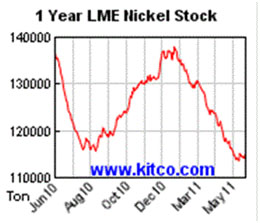 China is the leading consumer of nickel and is competing for supplies with recovering U.S. industrial demand, as well as India, Russia and Brazil.
China is the leading consumer of nickel and is competing for supplies with recovering U.S. industrial demand, as well as India, Russia and Brazil. The global automotive industry is using more and more nickel for electric and hybrid vehicles.
Mining companies have shifted exploration efforts to more challenging locations like the Arabian Peninsula, east-central Africa and the Subarctic because of a lack of discovery of new sulfide deposits in traditional mining camps.
The largest nickel-producing companies, Norilsk, Vale and Xstrata, don't actually provide a lot of leverage to nickel, as they are diversified minersmeaning they also produce iron, manganese, bauxite, aluminum, copper, coal, cobalt, potash, platinum group metals, gold and silver.
Pure nickel exploration plays aren't common. Are there one or two junior nickel plays on your radar screen?
If not maybe there should be.
Richard (Rick) Mills
[email protected]
www.aheadoftheherd.com
If you're interested in learning more about resource, bio-tech and technology companies please come and visit us at www.aheadoftheherd.com.
Free investors forum, free newsletter, no credit card or personal information is asked for.
Richard is host of Ahead of the Herd and invests in the junior resource sector. His articles have been published on over 300 websites, including: The Wall Street Journal, The Gold/Energy Reports, Calgary Herald, USA Today, National Post, Financial Sense Vancouver Sun, Forbes, Huffington Post, SafeHaven, Market Oracle, Stockhouse, Lewrockwell, Casey Research, 24hgold, SilverBearCafe, Infomine, Mineweb, 321gold, Kitco, Gold-Eagle, Resource Investor, FNArena and Metals News.
Legal Notice/Disclaimer
This document is not and should not be construed as an offer to sell or the solicitation of an offer to purchase or subscribe for any investment. Richard Mills has based this document on information obtained from sources he believes to be reliable but which has not been independently verified.
Richard Mills makes no guarantee, representation or warranty and accepts no responsibility or liability as to its accuracy or completeness. Expressions of opinion are those of Richard Mills only and are subject to change without notice. Richard Mills assumes no warranty, liability or guarantee for the current relevance, correctness or completeness of any information provided within this Report and will not be held liable for the consequence of reliance upon any opinion or statement contained herein or any omission.
Further, I, Richard Mills, assume no liability for any direct or indirect loss or damage or, in particular, for lost profit, which you may incur as a result of the use and existence of the information provided within this Report.
Richard Mills does not own shares of any companies mentioned in this report.