Novavax Shares Shoot 65% Higher on Phase 3 COVID-19 Vaccine Trial's 89.3% Effectiveness
Source: Streetwise Reports (01/29/2021)
 Novavax Inc. shares established a new 52-week high after the firm's COVID-19 vaccine demonstrated an 89.3% efficacy rate in a Phase 3 trial in the U.K. and also showed efficacy against the South African variant in a Phase 2b trial.
Novavax Inc. shares established a new 52-week high after the firm's COVID-19 vaccine demonstrated an 89.3% efficacy rate in a Phase 3 trial in the U.K. and also showed efficacy against the South African variant in a Phase 2b trial.
read more >
Abiomed Shares Gain 8% After Firm Announces Record Q3 Revenue of $232 Million
Source: Streetwise Reports (01/28/2021)
 Shares of cardiac medical device maker Abiomed Inc. traded higher after the company reported record revenue for Q3/21 despite the impact from COVID-19.
Shares of cardiac medical device maker Abiomed Inc. traded higher after the company reported record revenue for Q3/21 despite the impact from COVID-19.
read more >
Sorrento Shares Rise 35% After Firm Posts Positive Data from Phase 1b ICU COVID-19 Trial
Source: Streetwise Reports (01/27/2021)
 Shares of Sorrento Therapeutics traded higher after the company released positive preliminary results from it of Phase 1b COVI-MSC Study for treatment of hospitalized ICU COVID-19 patients.
Shares of Sorrento Therapeutics traded higher after the company released positive preliminary results from it of Phase 1b COVI-MSC Study for treatment of hospitalized ICU COVID-19 patients.
read more >
Junior Mining Sector 'the Best Value in the Space'
Source: Streetwise Reports (01/27/2021)
 The StreetSmart Live! Broadcast on Jan. 26, 2021, focused on metals and why now is a good time for investors to be looking at them and the mining space. John Newell, independent analyst and portfolio manager, and John Feneck, mining sector expert and president of Feneck Consulting, addressed the status of the current metals market. CEOs of the three metals-focused companies comprising the Metallic Group of CompaniesGroup Ten Metals, Metallic Minerals and Granite Creek Copperdiscussed their projects.
The StreetSmart Live! Broadcast on Jan. 26, 2021, focused on metals and why now is a good time for investors to be looking at them and the mining space. John Newell, independent analyst and portfolio manager, and John Feneck, mining sector expert and president of Feneck Consulting, addressed the status of the current metals market. CEOs of the three metals-focused companies comprising the Metallic Group of CompaniesGroup Ten Metals, Metallic Minerals and Granite Creek Copperdiscussed their projects.
read more >
Vir Shares Climb 48% on Initial Phase 1 Chronic Hepatitis B Trial Data
Source: Streetwise Reports (01/26/2021)
 Vir Biotechnology shares traded higher after the company reported data from its Phase 1 Chronic Hepatitis B Study that showed VIR-3434 significantly and rapidly reduced hepatitis B surface antigen.
Vir Biotechnology shares traded higher after the company reported data from its Phase 1 Chronic Hepatitis B Study that showed VIR-3434 significantly and rapidly reduced hepatitis B surface antigen.
read more >
Aurinia Pharma Shares Open at 52-Week High as FDA Approves Adult Lupus Nephritis Drug
Source: Streetwise Reports (01/25/2021)
 Shares of Aurinia Pharmaceuticals traded 30% higher after the company reported that the U.S. Food and Drug Administration approved its LUPKYNIS (voclosporin) for use in treating adults with active lupus nephritis.
Shares of Aurinia Pharmaceuticals traded 30% higher after the company reported that the U.S. Food and Drug Administration approved its LUPKYNIS (voclosporin) for use in treating adults with active lupus nephritis.
read more >
AzurRx Doses First Two People in Expanded Phase 2b Trial of Cystic Fibrosis Patients
Source: Streetwise Reports (01/22/2021)
 AzurRx BioPharma shares traded 45% higher after the firm advised it had dosed the first two patients in its Phase 2b OPTION 2 Extension Study of MS1819 for treatment of exocrine pancreatic insufficiency in patients with cystic fibrosis.
AzurRx BioPharma shares traded 45% higher after the firm advised it had dosed the first two patients in its Phase 2b OPTION 2 Extension Study of MS1819 for treatment of exocrine pancreatic insufficiency in patients with cystic fibrosis.
read more >
NGL Energy Shares Rise 21% as It Seeks to Raise $2.05 Billion from Senior Secured Note Offering
Source: Streetwise Reports (01/21/2021)
 Shares of NGL Energy Partners LP traded higher after the firm made a series of announcements over the past three days regarding an update on financial operations, a settlement with a customer in bankruptcy, a quarterly Class D unit distribution and a $2.05 billion senior secured debt offering.
Shares of NGL Energy Partners LP traded higher after the firm made a series of announcements over the past three days regarding an update on financial operations, a settlement with a customer in bankruptcy, a quarterly Class D unit distribution and a $2.05 billion senior secured debt offering.
read more >
Explorer Discovers New Gold Mineralization at Abitibi Gold Belt Property
Source: Streetwise Reports (01/21/2021)
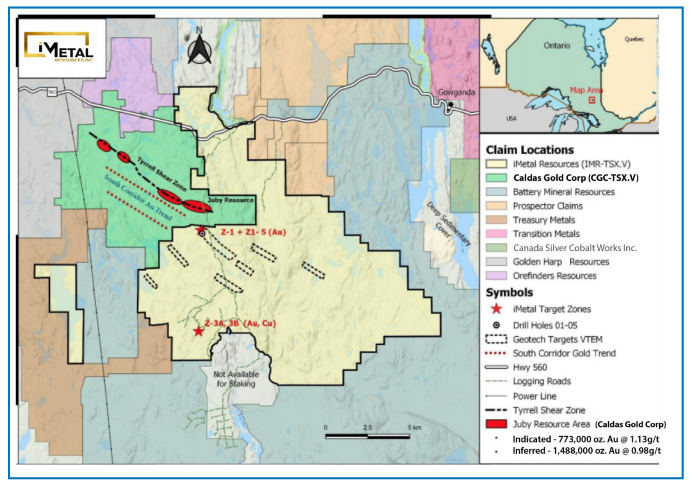 iMetal Resources is setting out to see if the multi-million ounce gold resource next door continues onto its Gowganda West property in Ontario.
iMetal Resources is setting out to see if the multi-million ounce gold resource next door continues onto its Gowganda West property in Ontario.
read more >
Gritstone Oncology Licenses LNP Platform from Genevant Sciences to Develop COVID-19 Vaccine
Source: Streetwise Reports (01/20/2021)
 Shares of Gritstone Oncology reached a new 52-week high price after the company advised it entered into a non-exclusive licensing agreement with Genevant Sciences to develop and commercialize self-amplifying RNA vaccines for use against SARS-CoV-2.
Shares of Gritstone Oncology reached a new 52-week high price after the company advised it entered into a non-exclusive licensing agreement with Genevant Sciences to develop and commercialize self-amplifying RNA vaccines for use against SARS-CoV-2.
read more >