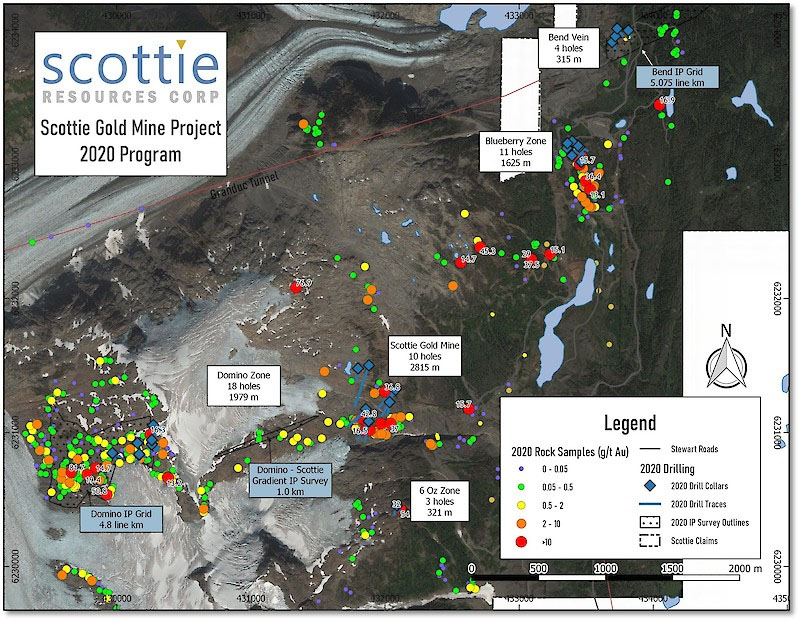
Lithium Batteries that Recharge in Seconds
Source: Technology Review, Katherine Bourzac (4/11/11)
"A new way of making battery electrodes has been used to create an extra fast-charging lithium-ion battery."
A new way of making battery electrodes based on nanostructured metal foams has been used to make a lithium-ion battery that can be 90% charged in two minutes. If the method can be commercialized, it could lead to laptops that charge in a few minutes or cell phones that charge in 30 seconds.
The methods used to make the ultrafast-charging electrodes are compatible with a range of battery chemistries; the researchers have also used them to make nickel-metal-hydride batteries, the kind commonly used in hybrid and electric vehicles.
How fast a battery can charge up and then release that power is primarily limited by the movement of electrons and ions into and out of the cathode, the electrode that is negative during recharging. Researchers have been trying to use nanostructured materials to improve the process, but there's usually a trade-off between total energy storage capacity (which determines how long a battery can run before needing a recharge) and charge rates.
Braun's group has made highly porous metal foams coated with a large amount of active battery materials. The metal provides high electrical conductivity, and even though it's porous, the structure holds enough active material to store a sufficient amount of energy. The pores allow for ions to move about unimpeded.
"It's some distance to a product, but we have pretty good lab demos" with nickel-metal-hydride and lithium-ion batteries, says Braun. Braun acknowledges: "There are lots of people coming up with elegant [electrode] structures, but manufacturing them is tricky." He says, however, that his fabrication process combines existing methods that are currently widely used to make other products, and that it shouldn't be too difficult to adapt them. The process would add extra steps to making a battery, but these steps aren't particularly expensive or complex, Braun says.
The methods used to make the ultrafast-charging electrodes are compatible with a range of battery chemistries; the researchers have also used them to make nickel-metal-hydride batteries, the kind commonly used in hybrid and electric vehicles.
How fast a battery can charge up and then release that power is primarily limited by the movement of electrons and ions into and out of the cathode, the electrode that is negative during recharging. Researchers have been trying to use nanostructured materials to improve the process, but there's usually a trade-off between total energy storage capacity (which determines how long a battery can run before needing a recharge) and charge rates.
Braun's group has made highly porous metal foams coated with a large amount of active battery materials. The metal provides high electrical conductivity, and even though it's porous, the structure holds enough active material to store a sufficient amount of energy. The pores allow for ions to move about unimpeded.
"It's some distance to a product, but we have pretty good lab demos" with nickel-metal-hydride and lithium-ion batteries, says Braun. Braun acknowledges: "There are lots of people coming up with elegant [electrode] structures, but manufacturing them is tricky." He says, however, that his fabrication process combines existing methods that are currently widely used to make other products, and that it shouldn't be too difficult to adapt them. The process would add extra steps to making a battery, but these steps aren't particularly expensive or complex, Braun says.